
The GSI Helmholtz Centre for Heavy Ion Research (GSI) in Germany got the upper hand as they changed their approach to “cold fusion:” focusing a low-energy beam at a target with a high atomic number led to the discovery of elements from bohrium (107) to copernicium (112). Every few years, a new element would be discovered and named, eventually reaching element 106 (seaborgium). and Russia, mainly the Lawrence Berkeley National Laboratory and the Joint Institute for Nuclear Research in Dubna, pushed forward in the 1950s, 60s, and 70s. Fittingly, the first element created this way was named mendelevium. The aim: to add a proton and increase the atomic number, thereby making a new element. If everything went exactly right, the nuclei of the atoms in the beam and the target fused. Scientists used cyclotrons and accelerators to bring ions of lighter elements to high speed, then fired them at the nuclei of elements with higher atomic numbers. Instead of brute force, finesse was the key. Beyond element 100, fermium, however, not even hydrogen bombs were powerful enough to produce new elements, so scientists changed their tactics. 
In radioactive material, physicists found new elements: neptunium, plutonium, americium, einsteinium, and more. Until physicists probed around in the debris produced in nuclear reactors and explosions, only 92 naturally occurring elements were known, up to uranium. This, Scerri writes in a 2013 article for Scientific American, “might undermine the very rationale behind the table’s existence: the recurring patterns that give the periodic table its name.” Exactly how many elements are still to be discovered? Is there an end to the periodic table? When will we reach it? What does it teach us about the nature of the elements? Creating New Elements Already, as atomic numbers reach ever higher, the chemical properties of some of the new elements do not resemble those of other elements in the same group. This law is being pushed to its limits as chemists synthesize new elements. Menshutkin while he went off to inspect cheese-making cooperatives.” But this changed quickly, “by 1871, Mendeleev was quite clear on his belief that he had isolated a new law of chemistry.” From the 1st English edition of Dmitrii Mendeleev’s Principles of Chemistry (1891) via Wikimedia Commons “Had Mendeleev been cognizant of the implications of the periodic system, he would likely not have relegated its presentation to the Russian Chemical Society in March 1869 to N.A. In the beginning, not even Mendeleev recognized the magnitude of his discovery, writes the historian of science Michael Gordin. Mendeleev didn’t just predict the existence of then-unknown elements, but also their properties. 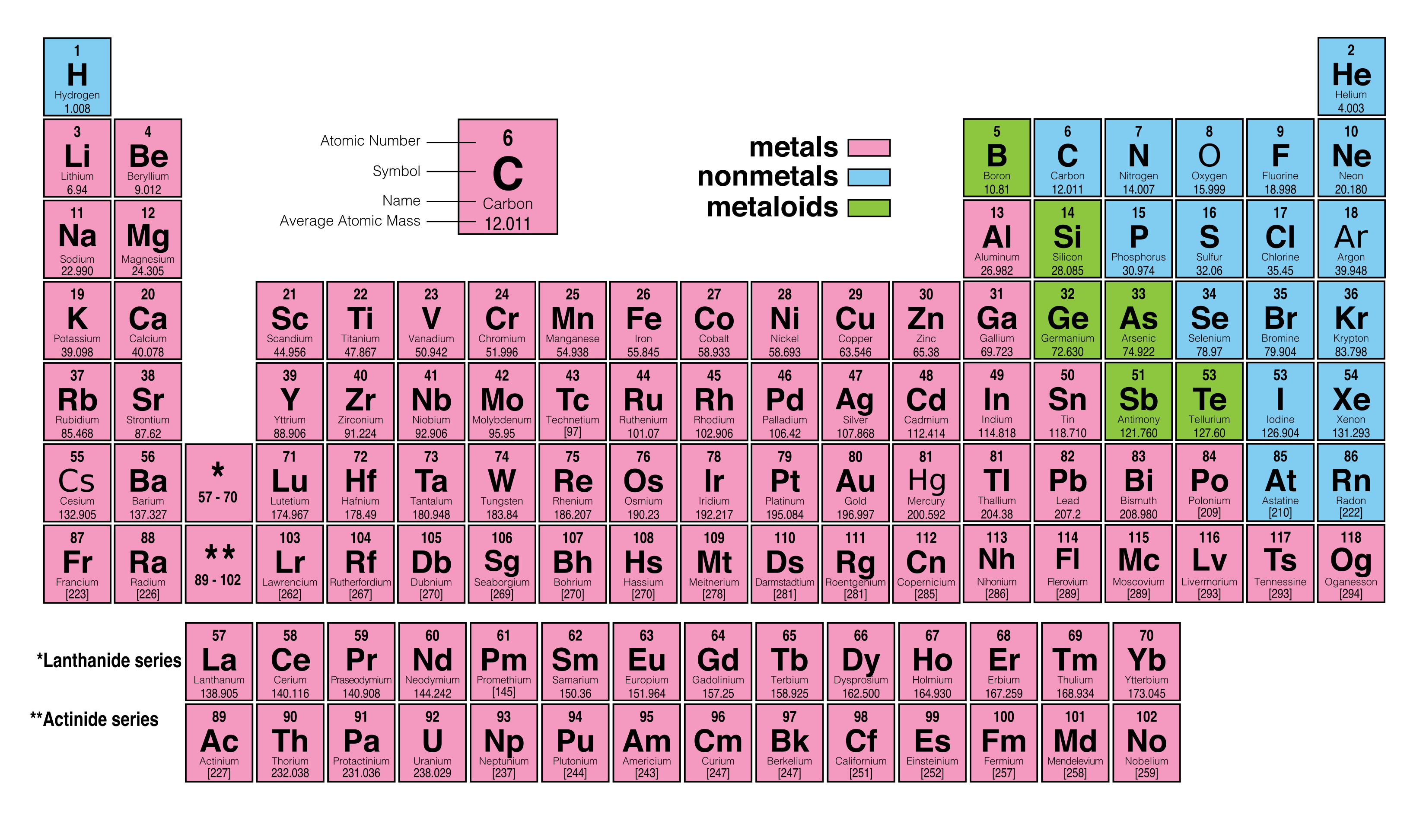
Soft metals like lithium and potassium, which react strongly with others, inhabit one column, while fluorine and iodine, non-metallic reactive elements, live in another. This configuration usually dictates an element’s “personality” as well as its size and shape. Mendeleev wasn’t the first to come up with a system to categorize elements, but, Scerri points out, “his version is the one that had the biggest impact on the scientific community.” The periodic table organizes elements by rows according to their atomic number, the number of protons in an atom’s nucleus, and by columns according to the configurations of the atom’s outermost electrons. In the beginning, not even Mendeleev recognized the magnitude of his discovery. It captures the essence of chemistry in one elegant pattern.” Dmitri Mendeleev first published a scheme to organize all then-known elements in 1869, and this system, although not perfect, became fundamental in the study of chemistry. “It is one of the most powerful icons of science. “It graces the walls of lecture halls and laboratories of all types, from universities to industry,” he wrote in American Scientist. The periodic table of elements is a “stalwart symbol” of chemistry, the chemist Eric R. Efforts to find the next elements, 119 and 120, are underway. The new elements also received their final names: nihonium, moscovium, tennessine, and oganesson. But as we enter the International Year of the Periodic Table, the classic periodic table has been filled to its seventh row: In late 2015, the International Union of Pure and Applied Chemistry officially confirmed elements 113, 115, 117, and 118. Until December 2015, there were holes in the periodic table, elements synthesized but not yet officially recognized.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
